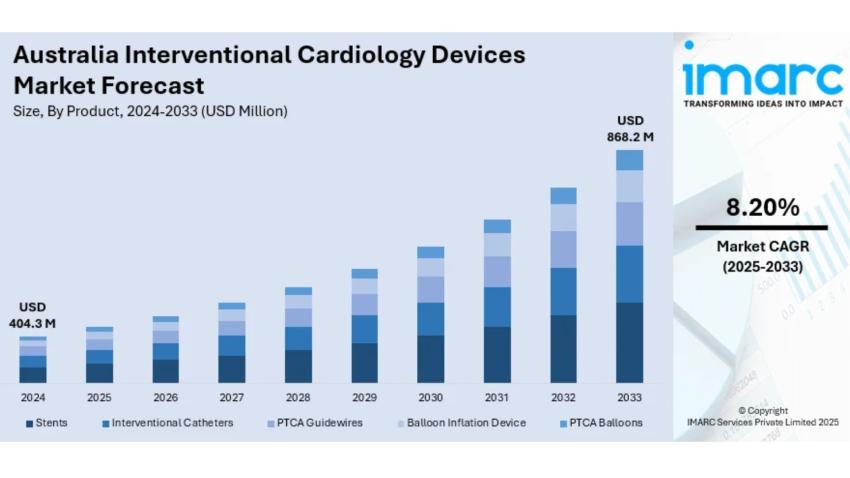
The latest report by IMARC Group, titled “Australia Interventional Cardiology Devices Market: Industry Trends, Share, Size, Growth, Opportunity and Forecast 2025–2033,” offers a comprehensive analysis of the Australia interventional cardiology devices market’s growth. The report also includes competitor and regional analysis, along with a breakdown of segments within the industry. The Australia interventional cardiology devices market size reached USD 404.3 Million in 2024. Looking forward, IMARC Group expects the market to reach USD 868.2 Million by 2033, exhibiting a growth rate (CAGR) of 8.20% during 2025–2033.
Report Attributes:
- Base Year: 2024
- Forecast Years: 2025–2033
- Historical Years: 2019–2024
- Market Size in 2024: USD 404.3 Million
- Market Forecast in 2033: USD 868.2 Million
- Market Growth Rate 2025–2033: 8.20%
For an in-depth analysis, you can refer to a sample copy of the report:
https://www.imarcgroup.com/australia-interventional-cardiology-devices-market/requestsample
Australia Interventional Cardiology Devices Market Overview
- The market is growing steadily because of an older population and more people having heart problems, which is making people look for better treatment options.
- Doctors are using less invasive procedures more often to help patients feel better faster and recover quicker.
- Hospitals and clinics are using smart tools powered by AI to watch and check heart conditions, which helps find irregular heartbeats and rhythm issues more accurately.
- More doctors are using drug-eluting stents because they help stop the arteries from narrowing again and keep the blood vessels open longer.
- New rules and approvals from groups like TGA are making it easier for new and better medical devices to be used quickly.
Key Features and Trends of Australia Interventional Cardiology Devices Market
- AI-driven cardiac monitoring tools, like the Medtronic Reveal LINQ with AccuRhythm, are improving the accuracy of heart condition diagnoses.
- More approvals from the TGA for new types of drug-coated stents and flexible delivery systems are changing how heart procedures are performed.
- Companies in the market are adding products such as advanced intravascular imaging and pressure-guiding wires to their offerings.
- Hospitals and clinics are spending more on digital workflow systems and remote monitoring tools to make patient care more efficient.
- Training and certification programs for heart specialists are helping to improve the quality of procedures and ensure safer patient outcomes.
Growth Drivers of Australia Interventional Cardiology Devices Market
- More people are getting heart diseases like coronary artery disease and other cardiovascular issues.
- There is a growing need for less invasive and quicker recovery treatments.
- More use of AI and smart tools for diagnosing heart problems is happening.
- Government policies, rules, and financial support for treatments are helping the field.
- New developments in stents, catheters, and other tools used in heart procedures are continuing.
Innovation & Market Demand of Australia Interventional Cardiology Devices Market
- AI-powered tools for monitoring and diagnosing are being used to automatically detect irregular heartbeats and make hospital processes more efficient.
- Companies are introducing newer types of drug-coated stents that are more flexible and work better for different groups of patients.
- Hospitals are using real-time data sharing and remote heart care systems to help with heart treatments in distant or rural areas.
- Working together with international medical equipment makers is helping bring advanced tools and software faster to local markets.
- More people are asking for treatments that are less invasive and quicker recovery programs for heart issues.
Australia Interventional Cardiology Devices Market Opportunities
- Expansion of AI and telemedicine presents opportunities for remote diagnosis and patient management, especially in regional Australia.
- The rise in complex coronary cases is fueling demand for intravascular imaging, FFR (fractional flow reserve) tools, and personalized device selection.
- Ongoing government support for digital health infrastructure is creating room for innovation in smart and wearable cardiac devices.
- Commercial partnerships with device manufacturers and MedTech startups are opening channels for product co-development.
- Investment in workforce development and specialist training is supporting higher procedure volumes and skill enhancement.
Australia Interventional Cardiology Devices Market Challenges
- Navigating stringent TGA regulatory compliance and product registration processes is a market barrier.
- Addressing device cost pressures and supporting affordable access in public healthcare settings.
- Managing supply chain disruptions for high-tech devices and components.
- Ensuring interoperability of new technologies with hospital EMRs and clinical systems.
- Bridging the skills gap among interventional cardiologists for newly introduced device technologies.
Australia Interventional Cardiology Devices Market Analysis
- The sector is dominated by leading global device manufacturers, with strong presence and competition in stents, balloons, and accessory devices.
- Product innovation is focused on flexible, imaging-guided, and drug-eluting platform development.
- Market segmentation highlights rapid adoption in coronary stents, balloon catheters, intravascular imaging, and accessory devices.
- Demand is highest in major urban and metropolitan cardiac centers, with uptake growing in regional markets as digital health expands.
- The hospital segment is the primary end user, emphasizing procedural volumes and investment in new technology.
Australia Interventional Cardiology Devices Market Segmentation:
- By Product:
- Coronary Stents (drug-eluting, bare-metal, bioresorbable)
- PTCA Balloon Catheters
- Intravascular Imaging Catheters and Pressure Guidewires
- Accessory Devices
- By Application:
- Coronary Artery Disease
- Structural Heart Disease
- Others
- By End User:
- Hospitals
- Specialty Clinics
- Ambulatory Surgical Centers
- By Region:
- Australia Capital Territory & New South Wales
- Victoria & Tasmania
- Queensland
- Northern Territory & Southern Australia
- Western Australia
Australia Interventional Cardiology Devices Market News & Recent Developments
- September 2024: SMT’s Supraflex Cruz drug-eluting stent received TGA approval, introducing a flexible, clinically proven option for interventional treatment of coronary artery disease.
- May 2024: Medtronic launched AI-enabled AccuRhythm algorithms in Reveal LINQ cardiac monitors, enhancing diagnostic accuracy for arrhythmia and AFib management in Australia.
Australia Interventional Cardiology Devices Market Key Players
- Abbott Laboratories
- Medtronic PLC
- Boston Scientific Corp
- Koninklijke Philips NV
- Cardinal Health Inc
- Terumo Corp
- Teleflex Inc
- B. Braun
- Biosensors
- BIOTRONIK
- Asahi Intecc Co Ltd
- Cook Medical
- Merit Medical Systems Inc
- SMT
- Zeon Corp
- Alvimedica
- Meril Life
- Medinol
Key Highlights of the Report
- Market Performance (2019–2024)
- Market Outlook (2025–2033)
- COVID-19 Impact on the Market
- Porter’s Five Forces Analysis
- Strategic Recommendations
- Historical, Current, and Future Market Trends
- Market Drivers and Success Factors
- SWOT Analysis
- Structure of the Market
- Value Chain Analysis
- Comprehensive Mapping of the Competitive Landscape
Note: If you need specific information that is not currently within the scope of the report, we can provide it to you as a part of the customization.
Ask analyst for your customized sample:
https://www.imarcgroup.com/request?type=report&id=33506&flag=E
FAQs: Australia Interventional Cardiology Devices Market
Q1: What is the projected value of the Australia interventional cardiology devices market by 2033?
A: The market is expected to reach USD 868.2 Million by 2033, growing at a CAGR of 8.20% during 2025–2033.
Q2: What factors are driving growth in the Australian interventional cardiology devices market?
A: Growth is driven by a rising aging population, growing prevalence of heart disease, advancement in minimally invasive technologies, and consistent regulatory support.
Q3: What are the leading device types in the Australian market?
A: Drug-eluting stents, AI-powered cardiac monitoring devices, balloon catheters, and advanced accessory devices like guidewires and imaging catheters.
Q4: What recent innovations are influencing the market?
A: TGA approval for new drug-eluting stents and AI-driven upgrades to implantable cardiac monitors are key developments.
Q5: Who are the major players in the Australian interventional cardiology devices market?
A: Key companies include Abbott, Medtronic, Boston Scientific, Philips, Terumo, and several others.
About Us:
IMARC Group is a leading market research company that provides management strategy and market research worldwide. We partner with clients in all sectors and regions to identify their highest-value opportunities, address their most critical challenges, and transform their businesses. Our solutions include comprehensive market intelligence, custom consulting, and actionable insights to help organizations make informed decisions and achieve sustainable growth.
Contact Us:
134 N 4th St. Brooklyn, NY 11249, USA
Email: sales@imarcgroup.com
Tel No: (D) +91 120 433 0800
United States: +1-631-791-1145




